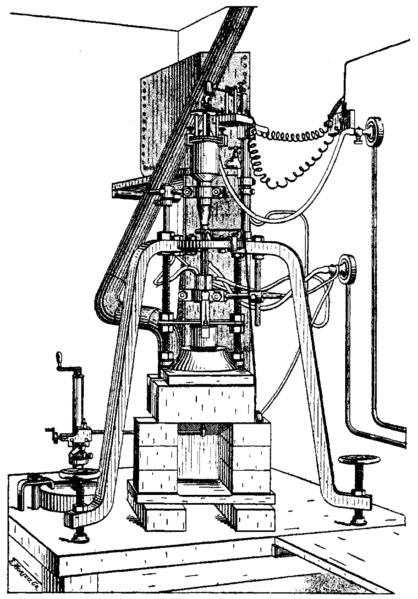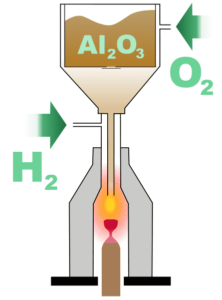
Flame fusion, discovered in 1902 by Auguste Verneuil, was the first successful process of creating synthetic gemstones. Using powdered elements and high temperatures he was able to grow crystals in a laboratory. Initially, rubies were the only gems produced by this method. Eventually, it was determined that by altering the elements put into the melt, other gems could be produced. In the 1940s Union Carbide used this process to create the famous “Linde Star” sapphires.
The process works as follows: powdered aluminum oxide travels towards a 2000-degree flame, the heat is created by that flame melts the powder, and droplets are formed. These droplets then fall onto a slowly turning earthen rod and, upon cooling, crystallize into a boule. The outside looks like a cylinder but inside, the newly created synthetic gem material has the crystal structure of the natural gem material. Color can be induced by adding certain chemical elements to the aluminum oxide powder mix. Chromium adds red to create ruby and titanium and iron combine to make blue sapphires. Red and blue are the two most-produced examples but just about any color can be created with this method. An alternate name for this process is Verneuil after its creator.